Title: Systematic band gap tuning BaSnO3 via chemical substitutions: The role of clustering mixed-valence perovskites.
Authors: Seugnhun Lee, Haihang Wang, Priya Gopal, Jongmoon Shin, H.M. Iftekhar Jaim, Xiaohang Zhang, Se-Young Jeong, Demet Usanmaz, Stefano Curtarolo, Marco Fornari, Marco Buongiorno Nardelli, and Ichior Takeuchi.
Publication Journal and Link: Chemistry of Materials, DOI: 10.1021/acs.chemmater.7b03381
Diamond, graphite, and graphene. What do these three materials have in common? Carbon! All three materials only contain carbon, but the way the carbon atoms are arranged in these materials are all different! And these differences in atomic arrangements have a drastic impact on the physical properties of these materials. For example, diamond is really hard because the carbon atoms are sp3-hybridized and covalently bonded to each other, whereas graphite cleaves rather easily due to the sp2-hybridized carbon atoms forming sheets that interact through weak intermolecular interactions.
Measuring materials properties and understanding that behavior at an atomic scale is an important task carried out by the materials chemist. If the materials chemist could determine the factors that govern a range of physical and chemical properties, there is an exciting opportunity for the targeted synthesis of a new material that displays a desired property. This could possibly shorten the development pipeline for a range of technologies, from room-temperature superconductors to drug delivery devices to thermochromic windows.
One material that has captivated the interests of materials chemists is BaSnO3, a material that could possibly be implemented in transparent conductors, dye-sensitized solar cells, and room temperature superconductors. Many researchers have studied this material, finding its physical properties can be tailored by introducing chemical substitutions. Both Ba and Sn could be replaced with other metals, alterations that typically result in changes in the electronic structure of the material. This ultimately manifests as an ability to tune the band gap of BaSnO3, or the wavelengths of light that BaSnO3 is capable of absorbing. The band gap is a critical physical property with implications on conductivity and transparency, as well a host of other characteristics. In this article, the researchers aimed to study the effect of substituting Sr, Pb, and Bi into the crystal structure of BaSnO3.
Thin films of pure and substituted BaSnO3 were synthesized via pulsed laser deposition, a technique that allows for the rapid production of a wide variety of materials with minor differences. The number of substitutions made (in mol %) ranged from 0% to slightly over 50% and computational studies were employed to model and verify these materials.
Upon substituting in Sr, the researchers determined that the lattice constant of BaSnO3 decreased with an increase in Sr substitution, implying that the smaller Sr atom was replacing the much larger Ba atom (Figure 1). This structural change caused the band gap of the material to increase with a rise in Sr concentration. After synthesizing a range of Pb-substituted BaSnO3, the authors found that Pb2+ cations substituted for Ba, while Pb4+ substituted for Sn. This led to a fascinating observation that the band gap shrank as Pb concentrations rose to 20 mol %, but expanded once Pb concentrations exceeded this threshold percentage. When the researchers synthesized Bi-substituted BaSnO3, they found that Bi ions didn’t substitute for any of the atoms in the lattice! Instead, computational studies suggested that the Bi atoms would cluster together. Indeed, the alterations in the band gap and light-absorbing characteristics verify this.
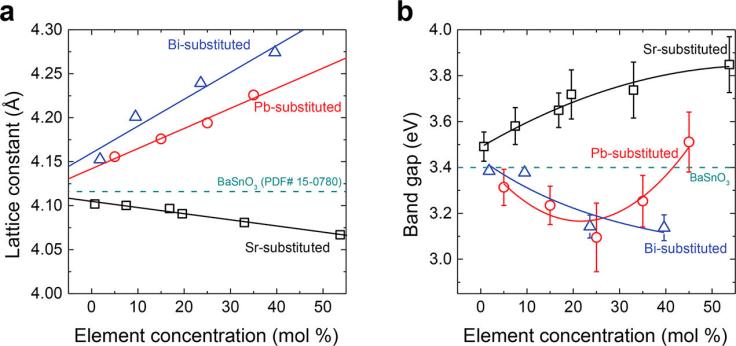
Figure 1. Substituting Sr, Bi, and Pb into BaSnO3 resulted in changes in (a) structural and (b) electronic changes.
This study into the changing physical properties of BaSnO3 as a function of substituting Sr, Pb, and Bi is a fascinating venture in the world of materials chemistry. Chemists can change how a material behaves by simply changing a small percentage of the atoms in the material. Here, researchers found that substituting in Sr, Pb, and Bi affect the chemistry of BaSnO3, with implications on characteristics associated with conductivity and absorption of light. These property changes were rooted in structural alterations at the atomic level. This work therefore serves as a roadmap for future tailoring of BaSnO3 for specific targeted applications.

Leave a comment